BRIEF DESCRIPTION OF PROJECTS
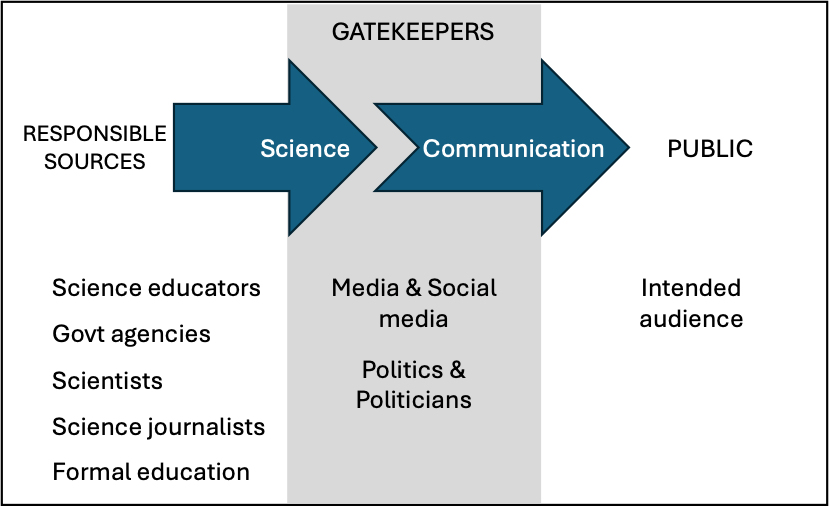
Science Communication – STEM students’ and instructors’ role identity perceptions. Recognition of the critical role of science communication in the public’s understanding of science has evolved steadily over the past few decades (Nisbet & Scheufele, 2009). However, the Covid-19 pandemic taught us—the hard way—that effective science communication requires more than a set of good presentation practices. This crisis made the impact of science communication on public beliefs and behavior more evident and immediate. Stakes were higher globally as well as at the most personal level. Scientists’ engagement with non-scientists reached a new level of shared awareness within the scientific community, thereby underscoring several key questions: Who is responsible for communicating science to the public? What attributes make science communication effective? Where and how should scientists receive training in science communication? What should be the ultimate goal of science communication?
The social role of modern scientists increasingly requires scientists to communicate effectively with the public. Yet, little research has examined how this expectation is reflected in formal chemistry education, professional training, or scientific practice. Similarly, limited attention has been given to how scientists perceive themselves in the role of science communicators (role identity). Our preliminary research reveals dissonances between the idealized and perceived models of science communication held by STEM students and instructors. In their idealized model, they see themselves as central figures in public science discourse; however, they perceive other agents’ influence (e.g. politicians, influencers) to be more significant. In addition, despite acknowledging a responsibility to communicate, many scientists possess only a rudimentary understanding of the nature of science communication and lack knowledge of effective strategies beyond generic performative skills (e.g. presentation skills). These gaps present significant obstacles to meaningful engagement with the public.
Our current research draws from our previous work examining how chemistry instructors’ self-image influences their teaching practices and identity as scientists. We argue that understanding how a communication role identity develops—and the factors that shape it—can lead to more effective, socially responsive training in science communication. We anticipate that reflective environments encouraging scientists to consider not just what and how to communicate, but also why to communicate, will better align science communication practice with current societal expectations.
Our experience researching chemistry graduate students’ role identity as instructors and how it informs their growth as scientists parallels our current science communication interests.
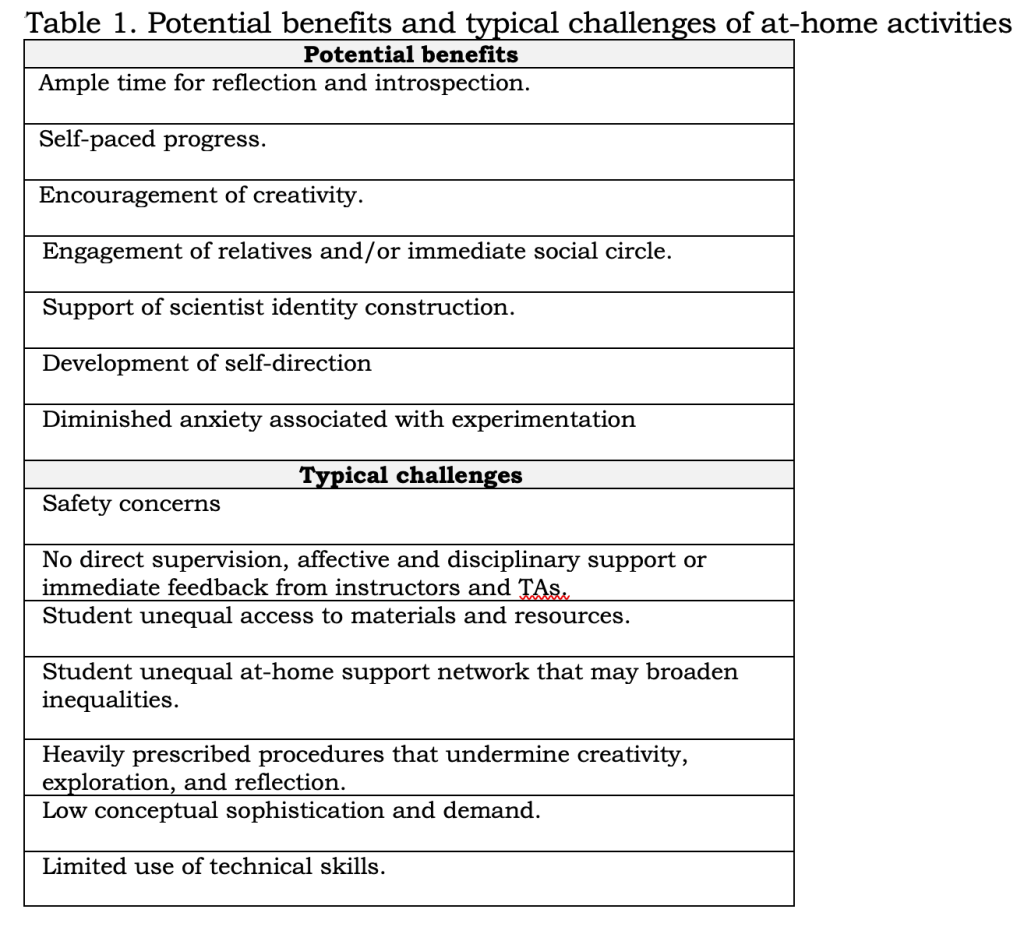
Experimentation activities away from the laboratory facilities. Pre-Covid, we explored the use of experimentation activities away from the laboratory facilities. We envisioned this approach would potentially provide an environment where students could be more reflective of their own work and learning and be more creative. Experimental activities away from the laboratory potentially alleviate some of the chemistry anxiety and work as induction to experimentation in the affective dimension, Table 1. shows benefits and challenges we considered for the selection and design of the activities. Four pillars guided the design of these activities: (1) safety, (2) exclusive use of household materials, (3) green chemistry principles, and (4) access to an authentic experimentation experience.
We envisioned an additional broad-scope benefit of these activities. About two-thirds of Costa Rican adults perceive science is little or not present in their daily lives (MICITT, 2012, p. 28). Science—and more probably so chemistry—is a distant and intangible concept with no immediate reference whose popular use is semantically void, especially for individuals who have attained only basic formal education. Through these homebound activities we want to invite students to engage their social and affective circle in learning about chemistry, the value of the profession, its applications and its ubiquity in daily life. Taking chemical experimentation out of the laboratory and vesting students as chemistry ambassadors in their homes and communities contributes to scientific literacy.
With the migration of courses to emergency remote teaching, ERT, due to COVID-19, we were presented with an otherwise unattainable unique scenario to test experimental activities away from the laboratory. Preliminary end-of-course survey data suggest the ERT course effectively supported development of nontechnical experimental skills and practices, e.g., asking scientific questions, planning and executing investigations, collecting, analyzing, and constructing explanations, creating arguments from evidence, and obtaining, evaluating, and communicating scientific information. These practices are relevant and transferable to the conventional course. Initial phenomenological analysis of participant in-depth interviews supports survey data and highlights the shortcomings of the learning environment. In-laboratory experiences are not substitutable; however, course designs may incorporate meaningful activities away from the lab to fulfill some of the objectives of experimental instruction assisting in optimization of instructional resources.
Multimodal Laboratories. Although research on learning in the tertiary-level college laboratory has surged, slow adoption of findings prevails. In fact, many take issue with the mere idea there is a need to move away from the traditional laboratory. Many factors contribute to this resistance. Amongst them is the impression that reform attempts to substitute rather than improve current experimental instruction, which results intimidating for often-pedagogically undertrained laboratory instructors. Furthermore, with few exceptions, research findings stem from sites with access to resources (researchers, doctoral students, infrastructure, and funding) that do not resonate with many end-consumers of research. Besides producing research evidence that may be more naturalistic to a larger portion of practitioners, we strive to propose an approach to reform that is simpler, more adaptable and less demanding. Multimodal labs use multiple modes of assignments, which in a broad sense–the experiment level–can be thought of as lab instructional styles, for example, those described by Domin (1999): expository, inquiry, discovery, and problem-based. Thus, practicals can range from carefully prescribed acid−base volumetric analysis (expository) to projects such as making an evidence-based recommendation between purchasing commercial calorimeters or utilizing Styrofoam cups (problem-based). Under a finer lens–the activity level–they refer to modes of engagement as described by Chi and Wylie(2014): passive (watching a demo of the use of a calorimeter), active (running a calorimeter simulation), constructive (integrating knowledge to develop a procedure to compare calorimeters), and interactive (dialogically engaging with peers to justify criteria to decide which of two calorimeters is preferable). We are currently probing the effectiveness of Multimodal Labs to assist students in developing significant learning through experimentation.
Characterization of successful experimental learning experiences using Langer’s Mindfulness Theory. We have used phenomenological approaches to study what happens in different laboratory instructional styles in an attempt to gain understanding of learning in this complex environment. Currently, we are using Ellen Langer’s Mindfulness Theory as framework to analyze qualitative data from our own studies and from research published over the past 20 years. Through this approach, we intend to capture empirical evidence that is diverse in terms of institution, student body, instructor expertise, and nature of intervention utilized. Characterization of these learning experiences with this unique framework will advance our understanding of what makes learning in the laboratory more effective.
Undergraduate Research. Our undergraduate research program strives to develop a research agenda constructed from the students’ perspectives and informed by their experiences as key participants in their own education. Findings may inform and impact undergraduate chemistry instruction. Currently, we are working on a study that intends to characterize conceptualization and use of Lewis structures by students across the chemistry curriculum and experts (faculty). The ultimate goal of this project is to propose a pedagogical approach to improve teaching/learning of this topic. In this project we have involved chemistry and science ed double majors. A second project looks at the implementation of Green Chemistry in the first year chemistry laboratory program. We introduce Green Chemistry as a new course topic but also as an innovative, modern way to conceptualize the practice of chemistry. The practicals are all reformed to align closer with Green Chemistry principles and the research goal is to assess how the curriculum affects entering students’ perception of chemistry as a socially responsible and environmentally sound endeavor.
Previous work. In the past we have carried out a series of studies to investigate the ‘self-explaining effect’ in large enrollment General Chemistry courses. The development and assessment of metacognition and its impact on chemistry problem solving continues to be central in our work. Our research uses both quantitative and qualitative inquiry approaches, as well as mixed methods designs. Following this line, we have developed in-class activities to be used in large enrollment courses to promote attention, participation, interest, and production of explanations all of which have been shown to benefit learning. We are assessing students’ perceived effect of such activities via qualitative and quantitative approaches.
The exploration of how students use online faculty rating sites and their perceived usefulness were among the initial ideas generated in consultation with the undergraduates. Besides assessment of instruction, undergraduates have focused on study habits of General Chemistry students, and participation of foreign-educated faculty in chemistry education in the US and its impact on curriculum. Recently, a group of students with a keen interest in History of Chemistry have investigated the occurrence of Multiple Discoveries in chemistry and the way they are presented in General Chemistry, as well as their potential as pedagogical tools. In a current study, undergraduates seek to characterize the use and conceptualization of Lewis structures by chemistry majors (novices) and instructors (experts). A cross-sectional approach furnishes qualitative and quantitative data of students at four stages in the program. In a second phase, this study intends to develop (design and pilot test) a pedagogical approach to effectively facilitate learning of the topic. This study contributes to international chemical education by analyzing evidence in light of findings drawn with student populations in other countries.